Heavy-Chain Disease
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
Heavy Chain Diseases (HCDs):
Clinical aspects and laboratory diagnostics
HEAVY CHAIN DISEASES (HCDs)
Rare, proliferative disorders affecting lymphoplasma B cells. They are characterized by the
presence of immunoglobulin heavy-chain fragments in the absence of free light chains.
MONOCLONAL GAMMOPATHIES: AN OVERVIEW
CLINICALLY OCCULT OR PRE-SYMPTOMATIC DISEASE FORMS
}
- MGUS
- Benign (inflammations, infections,...)
- Transitory (drugs, viral infections,...)
CLINICAL MANIFESTATIONS DUE TO PROLIFERATION OF NEOPLASTIC CLONES
}
- Multiple Myeloma
- Plasma cell leukemia
- Waldenström's macroglobulinemia
- α, γ and μ heavy chain disease
CLINICAL MANIFESTATIONS DUE TO PATHOLOGICAL EFFECTS OF MCs
}
- Chronic cold-agglutinin disease
- Amyloidosis
- Light-chain deposition disease
- Polyneuropathies
HCDs
HCDs can be considered as variants of
non-Hodgkin's lymphoma.
Three main types of HCDs
α-HCD: the most common form of the disease. It presents as extranodal marginal zone lymphoma of the lymph node mucosa-associated tissue;
γ-HCD or Franklin's disease, presents as non-Hodgkin lymphoplasmacytic lymphoma;
μ-HCD, the rarest of all variants, presents as small non-Hodgkin's lymphoma or chronic lymphocytic leukemia (CLL).
D L.Wahner-Roedler and R.A Kyle-Best Pract & Res Clin Haem Vol. 18,No.4, pp.729–746,2005
α HEAVY CHAIN DISEASES
Features
Geographically, patients affected by HCDs (approximately 400 by the end of the 70s) mainly reside in countries stretching from the Mediterranean area to the Middle Eastern countries
Prevalence is slightly higher in men than in women.
α-HCD mainly affects younger subjects, with ages ranging from 20 to 30 years.
STRUCTURES OF α-HCD PROTEINS
Most α-HCD proteins consist of multiple polymers.
The basic monomeric unit ranges from 29 to 34 kDa
Characteristics of a defective α chain:
missing V regions,
loss of CH1domains
absence of free light chains.
Most α-HCD proteins contain unusual N-terminus sequences.
Nonetheless, all α-HCD proteins analyzed fall under the α1 subclass group.
STRUCTURE OF DIFFERENT α-HCD PROTEINS
COMPARED TO THE PROTEIN STRUCTURE OF NORMAL HEAVY CHAINS
Beutler E et al. Williams Hematology 6th edn, pp 1327-1336; New York McGraw-Hill-2001
GENE SEQUENCES ENCODING FOR α-HCD PROTEINS
Show non-contiguous deletions in the V/J as well as in the S/CH1 regions.
Genomic structures with unusual coding regions display unusual sequences of different length, with coding and non-coding nucleotides.
The inserted regions show no homology with any known sequence!
STRUCTURAL ANALYSIS OF 3 GENES ENCODING FOR THE HCD PROTEIN: COMPARISON WITH THE NORMAL α1PROTEIN-CODING GENE
Beutler E et al. Williams Hematology 6th edn, pp 1327-1336; New York McGraw-Hill)-2001
CLINICAL FEATURES
Patients mainly present with digestive discomfort
The condition is characterized by malabsorption, manifested by:
diarrhea
weight loss
abdominal pain
·Amenorrhea, alopecia and growth retardation in children correlate with the extent and severity of disease progression
· Upon physical examination, these patients present with oedema, ascites and tetanus
LABORATORY INVESTIGATIONS
METABOLIC AND HEMATOLOGIC ABNORMALITIES
Anemia is often present.
Hypocalcemia, hypomagnesemia and hypoalbuminemia
Possible increase in alkaline phosphatase levels as a result of increased intestinal isoenzyme fractions.
Malabsorption detecting tests usually yield positive results.
PROTEIN ANALYSIS
· 50% of all cases show a detectable abnormal band in the β-globulin or in the α2 regions due to the polymerization of α-chains.
· The remaining 50% show normal serum protein electrophoretic patterns, without evidences of abnormal protein.
If the serum concentration is low, the pathological protein may escape detection by means of immunofixation!
THERAPY
Therapeutical schemes require:
· nutritional improvement
· reduction of exposure to intestinal pathogens
In the absence of hyper-growth of intestinal bacteria, the use of tetracyclines is deemed appropriate.
Good results have been achieved by means of cyclophosphamide, doxorubicin and prednisone treatment, as well as with vincristine, prednisolone, metatrexate, and dexamethasone.
Advanced stage disease should not exclude surgical treatment!!
DIAGNOSIS AND PROGNOSIS
The diagnosis of α-HCDs requires
the detection of an incomplete immunoglobulin heavy chain fragment in either serum or urine, unaccompanied by light chains.
The prognosis is variable and no specifically standardized treatment options are available.
Early stage α-HCD may respond to antibiotic treatment.
γ-HEAVY CHAIN DISEASES
Since the mid-60s, only 120 patients affected by γ-HCD have been reported worldwide in the literature.
Although γ-HCD affects all genders,
there is a clear predominance of female patients
The underlying causes of this disease are still unknown!
STRUCTURAL ABNORMALITIES OF THE PROTEINS
· Most γ-HCD proteins consist of incomplete heavy chain dimers, unbound to light chains
· The molecular weight of the monomeric unit ranges from 27 to 49 kDa
· Variable in length. They usually correspond to approximately 3/8 of the length of normal γ chains.
STRUCTURE OF DIFFERENT γ-HCD DEFECTIVE PROTEINS
COMPARED TO NORMAL γ-HEAVY CHAINS
Beutler E et al. Williams Hematology 6th edn, pp 1327-1336; New York McGraw-Hill-2001
STRUCTURAL FEATURES
Structural analysis of defective monoclonal γ heavy chains revealed the following characteristics:
· All proteins generally begin with a variable region.
· The short sequence is interrupted by a long deletion that includes the remaining portion of the V region.
· All γ-HCD proteins lack the entire CH1 domain. Thus, the normal sequence either begins with the hinge region H, or at the level of the CH2 domain.
GENE STRUCTURE OF DIFFERENT γ-HCD DEFECTIVE PROTEINS
COMPARED TO THE GENE STRUCTURE OF NORMAL γ HEAVY CHAINS
Beutler E et al. Williams Hematology 6th edn, pp 1327-1336; New York McGraw-Hill-2001
γ-HCDs
γ-HCDs fall into three main categories:
1) Disseminated lymphoproliferative diseases
Account for 60% of all cases
2) Localized proliferative diseases
Localized lymphoproliferative processes (25% of all cases)
3) Non-visible proliferative diseases
Patients with autoimmune disorders with or without lymphoproliferative processes.
LABORATORY INVESTIGATIONS
HAEMATOLOGICAL ABNORMALITIES
· Anemia is frequent
· Autoimmune hemolytic anemia has often been reported in association with thrombocytopenic purpura
· Lymphocytosis with chronic lymphocytic leukemia (CLL) may occur
· Circulating plasma cells or plasmacytoid lymphocytes have sometimes been observed
LABORATORY INVESTIGATIONS
Protein detection
Serum protein electrophoresis is not always capable of detecting the presence of a γ heavy chain MC
it may appear in the β1 or in the β2 regions
The diagnosis is established by means of serum immunofixation or by means of urine immunofixation using concentrated urine specimen
PROGNOSIS
The clinical course of a γ-HCD is extremely variable, ranging from totally asymptomatic and benign affections to rapidly progressing tumors, leading to death within weeks.
The total amount of serum γ-HCD protein correlates with the severity of the related maligancy.
The disappearance of the monoclonal component from serum and urine is associated with a complete apparent regression, induced by chemotherapy, radiation therapy or surgical removal of localized processes.

μ HEAVY CHAIN DISEASES
EPIDEMIOLOGY AND PATHOGENESIS
· μ-HCDs are extremely rare
· Since the first case was detected in 1969, only 33 other cases have been reported worldwide
· The average age at diagnosis is approximately 60 years (ranging between 15-80 years)
· The causes underlying μ-HCD are still unknown
STRUCTURAL ABNORMALITIES OF μ PROTEINS
· The molecular weight of μ-HCD proteins varies from 26.5 to 15.8 kDa, depending on the polymerization of μ chain fragments.
· As for other HCD types, the incomplete heavy chains of patients with μ-HCD contain long deletions of variable regions.
· Moreover, as opposed to other HCD proteins, μ-HCD proteins retain normal constant regions.
STRUCTURE OF DIFFERENT μ-HCD DEFECTIVE PROTEINS
COMPARED TO NORMAL μ HEAVY CHAINS
Beutler E et al. Williams Hematology 6th edn, pp 1327-1336; New York McGraw-Hill-2001
STRUCTURAL ANALYSIS OF A GENE ENCODING FOR A μ-HCD
PROTEIN: COMPARISON WITH THE NORMAL μ PROTEIN-CODING GENE
The gene encodes for a normal constant μ region, containing the CH1 domain. The VDJ region is present, but contains a single base deletion generating three stop codons.
A nucleotide sequence is inserted immediately after the 3' of the cleavage site in the destroyed J region, inducing the cell to cut the donor site at the 3 'end of the leading sequence, until the CH1 domain, thus eliminating the variable region sequences from mature mRNA.
CLINICAL FEATURES
Common symptoms of μ-HCD are typically related to the lymphoproliferative malignancy associated to lymphoplasmacytic cell proliferation, including chronic lymphocytic leukemia (CLL)
μ-HCD proteins have been reported in association with:
· SLE,
· liver cirrhosis
· hepatosplenomegaly with ascites
· lung infections
· splenomegaly
· myelodysplasia
LABORATORY INVESTIGATIONS
HEMATOLOGIC ABNORMALITIES
Frequent anemia. Rare lymphocytosis and thrombocytopenia.
Bone marrow examination typically shows increased amounts of lymphocytes as well as plasma cells and plasmacytoid lymphocytes.
PROGNOSIS
The clinical course of μ-HCD is quite variable.
The average life expectancy from diagnosis is 24 months.
THERAPY
There is no specific therapy for μ-HCD.
· The detection of a μ-HCD protein in serum should be regarded as an MGUS.
· Multiple drugs are used in combination, such as cyclophosphamide, vincristine and prednisone

Mercks index 2010
Ouchterlony
double radial immunodiffusion
· The antibody is dispensed into the central well. The antigens are distributed in the surrounding wells.
· Antibody and antigen diffuse in agar. An immunoprecipitate is formed when the equivalence point is reached. Coomassie blue (a specific protein stain) staining can be used in order to detect precipitation
· The experiment is carried out in moist chambers.
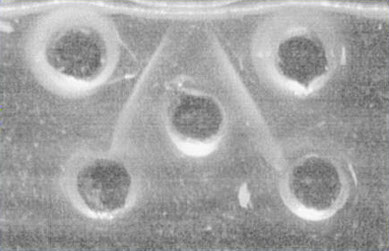
Tharp AM, Woodruff RD and Shihabi 7K
Annals of Clinical & Laboratory Science 33:97-100 (2003
Ouchterlony double radial immunodiffusion
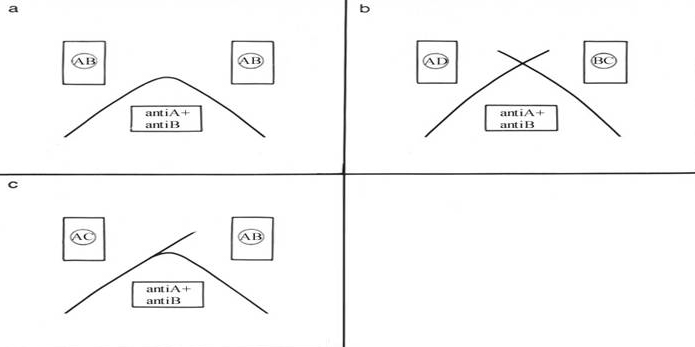
Diagrams of possible precipitation models of double immunodiffusion with one antiserum and 2 different antigen mixtures
A: identical epitopes
B: no common epitope
C: partial identity

CONCLUSIONS
· HCDs are rare diseases, not readily detectable among the variety of immunofixation patterns.
· Through a series of simple steps, this protocol allows to confirm the presence of rare immunofixation patterns. These are difficult to interpret, and may include the presence of incomplete Igs.
· IFE of supernatant enabled the identification of IgG3 in two cases, by using specific anti-IgG-subclass specific antisera.
· The technique is reproducible, and can easily be employed by labs that occasionally perform MC detection.
<
>