Introduction
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
The Diagnostic Approach

A biological sample is neither uniform nor can be considered to be "fully representative", since it is sampled from a population showing inter- and intra-individual biological variability.
Data from these samples cannot be interpreted directly, but requires a comparison to reference values.
CORRECT INTERPRETATION OF RESULTS:
• "distribution of results" relative to reference values
• result variability
Cvi (Intra-individual biological variability) is defined as the estimated amplitude of causal oscillations of values (around a homeostatic point), relative to each single analyte for every individual.
Cvg (Inter-individual biological variability) is defined as the variation between a set of homeostatic points, relative to the same analyte, within a group of individuals.
2 CAUSES OF VARIABILITY
TOTAL VARIABILITY
ANALYTICAL VARIABILITY
BIOLOGICAL VARIABILITY
PRE-ANALYTICAL
ANALYTICAL
POST-ANALYTICAL
INTRA-INDIVIDUAL
INTER-INDIVIDUAL
VARIABILITY
represents one of the main challenges physicians are faced with when dealing with the clinical use of laboratory data
BIOLOGICAL VARIABILITY
· INTRA-INDIVIDUAL VARIABILITY: within the same individual
· INTER-INDIVIDUAL VARIABILITY: among different individuals
MAIN SOURCES OF INTRA-INDIVIDUAL VARIABILITY
· Circadian Rhythm
· Seasonal changes
· Diet
· Menstrual Cycle
· Pregnancy
MAIN SOURCES OF INTER-INDIVIDUAL VARIABILITY
· Gender
· Age
· Race
· Body Mass
· Smoking
· Alcohol
· Drugs, Medications and Treatments
ANALYTICAL VARIABILITY
pre-analytical
post-analytical
analytical
VARIABILITY IN THE PRE-ANALYTICAL PHASE
· Sample collection and storing methods
· Sample labelling and patient identification
· Use of adequate material
· Use of analyte stabilizers
Critical points in patient management
Patients must be adequately instructed prior to blood collection.
General instructions for routine blood examinations:
· 12-hour fasting before blood collection
· Avoid physical exercise and stress 12 hours before tests
· Wait for the patient to be at rest before performing phlebotomy
Further recommendations to patients should be provided regarding the following examinations:
Clinical culture tests
Occult blood tests
Monitoring of patients undergoing oral anti-diabetic treatment
Monitoring of patients undergoing pharmacological treatments and therapies for diagnostic purposes
VARIABILITY IN THE ANALYTICAL PHASE
· Standardized procedures
· Internal controls
· Validation of unusually high results
VARIABILITY IN THE POST-ANALYTICAL PHASE
· Composition of a comprehensible clinical report
· Fast delivery of results
VENOUS BLOOD SAMPLING-RELATED ANALYTICAL VARIABILITY
BLOOD COLLECTION
Vacuum blood collection system
Syringe blood collection system
In vitro haemolysis
HAEMOLYSIS: confers a red hue to serum or plasma following sample centrifugation

PHYSICAL BARRIERS
Elimination of remixing risk
Physical separators between the liquid and the solid (coagulated) phases of serum or plasma, in order to favour their separation
Simplified sample transportation following centrifugation
1) pre-centrifugation integrated systems: pre-treated sampling tubes containing devices such as cylindric granules, polystyrene spheres or serum gel separator with the addition of granules.
2) pre-centrifugation integrated systems for blood tubes following blood collection: funnels preloaded with silicon mixtures
3) post-centrifugation systems: different types of filters
SAMPLE TRANSPORT TO THE LAB
Specimens should be processed immediately. Long processing times may affect material stability and test accuracy.
Within 45 minutes from blood collection → proceed with centrifugation and phase separation within 1 hour from collection
Test on coagulation factors → require immediate processing
Complete blood count → within 7 hours from collection
ESR → within 24 hours from collection
GLUCOSE: ↓↓↓ at all temperature (metabolized)
K+: ↑↑↑ due to erythrocyte and platelet lysis
Each sample should be fully labelled before it reaches the laboratory
The laboratory may choose to refuse such samples
· Patient Name and Surname should be indicated (ward, room and bed number should be specified in case of hospitalized patients)
· Labels should be applied to blood collection tube side (NEVER ON CAP). Waterproof ink should be used for labels.
· Date and time of collection should be reported on label
· unlabelled or inappropriately labelled samples
· samples damaged during transportation
· inappropriate sampling or storage
· samples contaminated by external material
SAMPLE STORAGE IN THE LABORATORY
Laboratories may need to store analyzed samples or specimens yet to be analyzed
COOLING (4°C)
Allows sample storage ranging from 2-3 days up to one week.
FREEZING (-20°C / -80°C)
Allows sample storage for longer periods. Ideal sample storage can be carried out by subdividing samples into small aliquots, immediately snap-frozen in liquid nitrogen prior to storage at -80°C.
Evaluating a diagnostic test
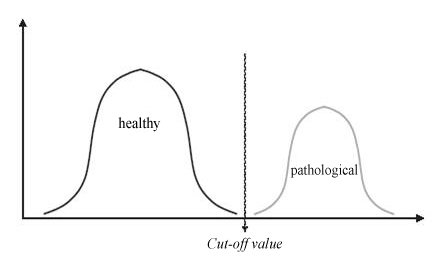
Reliability vs Validity
· Reliability: the ability of a test to guarantee reproducibility of results across repeated measurements
· Validity: the ability of a test to distinguish patients from healthy individuals within a population
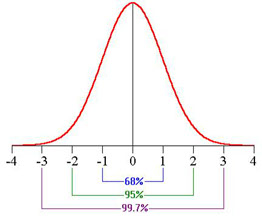
REFERENCE INTERVALS
A Gaussian distribution, with conventional limits of -2 and +2 Standard Deviations, implies that:
2,5% of observations will lie to the left of this interval
2,5% of observations will lie to the right of this interval
95% of observations will lie within this range
Evaluating Validity: the ideal scenario
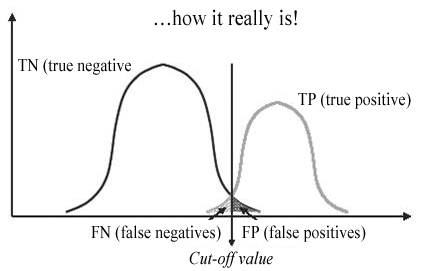
Evaluating Validity: the real scenario
Criteria of reliability
Sensitivity
Specificity
Dependent on method
Accuracy
Precision
Dependent on execution
Precision and Accuracy
Metrology: the discipline that studies physical measurements (theory of errors)
Precision: degree to which a set of data converges to the mean value or SD (not always true)
Accuracy: degree of closeness between the mean value and the true value
Instrumental accuracy: does not consider systematic errors
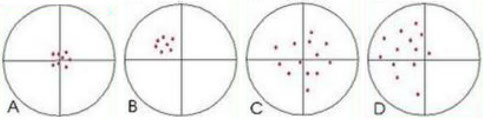
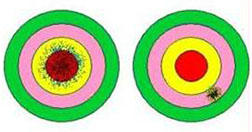
A) Precise and accurate - B) Precise, not accurate - C) Not precise, accurate - D) Not precise, not accurate
Slide30
Accuracy
Definition: degree of closeness between values obtained by means of analysis of a given parameter and the "true"

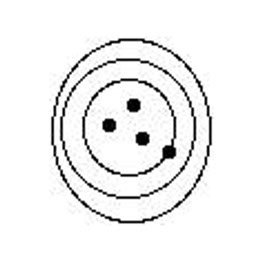
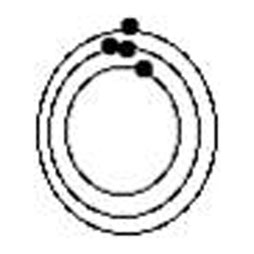
Precision
Definition: closeness of agreement between results obtained by repeated measurements of a given parameter within a defined sample
The evaluation of a new method's precision is usually obtained by assessing repeatability of measurements by performing replicate analyses and by calculating the range of variability.
RANGE OF REPRODUCIBILITY: refers to the range of variability. It defines significant variations in assay measurements.
Choosing and validating the most appropriate method within the context of in vitro diagnostics requires the assessment of two fundamental components:
SENSITIVITY and SPECIFICITY
ANALYTICAL SENSITIVITY
Minimum amount of analyte that a given method is able to detect
(least significant difference from 0).
ANALYTICAL SPECIFICITY
The ability of a method to specifically detect the analyte of interest
(minimizing interference level towards 0)
2 x 2 table
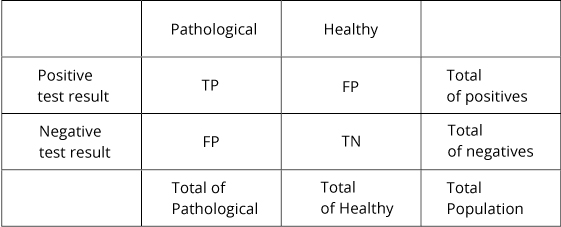
Sensitivity
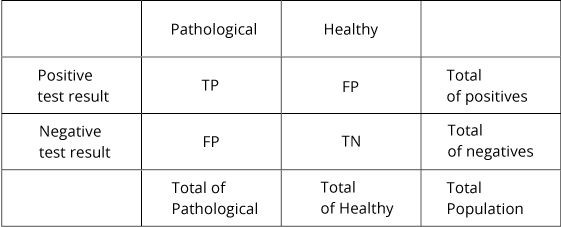
TP / Tot of Pathological: the ability of the assay to correctly identify pathological subjects.
Specificity
TN / Total of Healthy: the ability of the assay to correctly identify healthy subjects within a population.
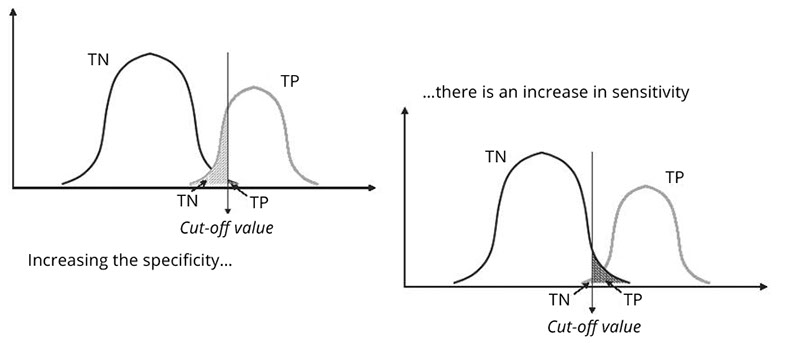
Sensitivity vs specificity
Predictive value of a test
How likely is it that a patient with a positive result (or a negative result) is truly pathological (or healthy)?
Positive Predictive Value (PPV)
VP / Total of positives: Indicates the likelihood that a subject is truly pathological given a positive test result.
Negative Predictive Value (NPV)
TN / Total of negatives: indicates the likelihood that a subject is truly healthy given a negative test result.
PV and prevalence
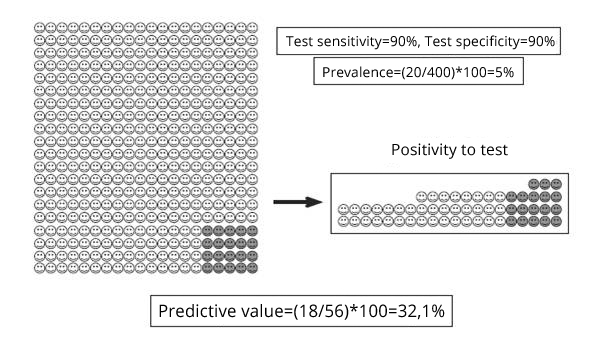
VP e prevalenza
Quality
· INTERNAL QUALITY CONTROLS
· EXTERNAL QUALITY CONTROLS
(at regional, national or international level)
COMMON MISTAKES
· incorrect sample labelling
· exchange of samples
· incorrect sampling procedure, inadequate sample storage
· incorrect execution of tests, miscalculated and misread results
· wrongly transcribed and/or assigned results
SYSTEMATIC ERROR
· incorrect concentration of reference solution (due to incorrect weighing procedures or the use of impure or expired material)
· incorrect instrument settings (wavelengths, pipettes, measuring cups)
· analytical methods lacking specificity
· human errors or mistaken operative procedures (incorrect choice of wavelength, incorrect calculations, systematic error associated to the measuring instrument)
RANDOM ERRORS
· inefficient or user "unfriendly" instruments
· contaminated or expired reagents
· inaccurate instrument measurements
· poor accuracy of instruments
· inadequate environment (due to dust, noise, temperature, light)
· unstable electric power supply
COMPOSITION OF A LAB REPORT
THIS IS A CRUCIAL AND HIGHLY RELEVANT ASPECT OF DIAGNOSTIC PROCEDURES
A LAB REPORT PROVIDES ANSWERS TO THE DIAGNOSTIC QUERIES POSED BY HEALTH SPECIALISTS
The value of lab report data lies in the clinical significance of such information in the context of patient management.
In other words, the real value of clinical laboratory tests derives from their practical utility for the patient.
· Should the effectiveness of laboratory activities be assessed by the production of a lab report or does it depend on the appropriate use of the report itself within a therapeutic diagnostic context?
· Answering this question implies the concept of "efficacy". No matter how accurately or rapidly a lab test result is generated, it is sterile unless:
a) it is analyzed, interpreted and followed by clinically relevant actions
b) it is implemented alongside other procedures that are critically relevant for patient management and for the choice of the therapeutic option.
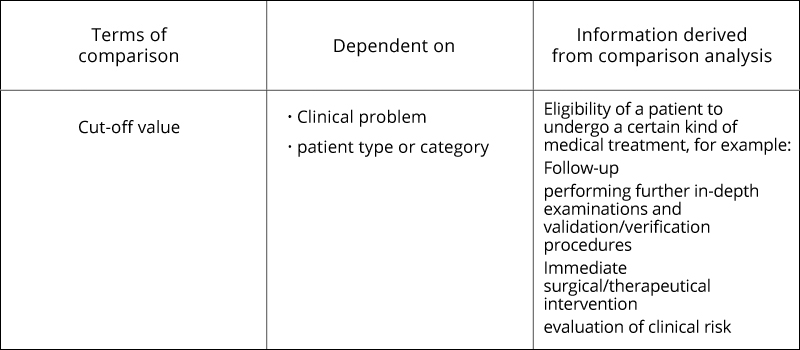
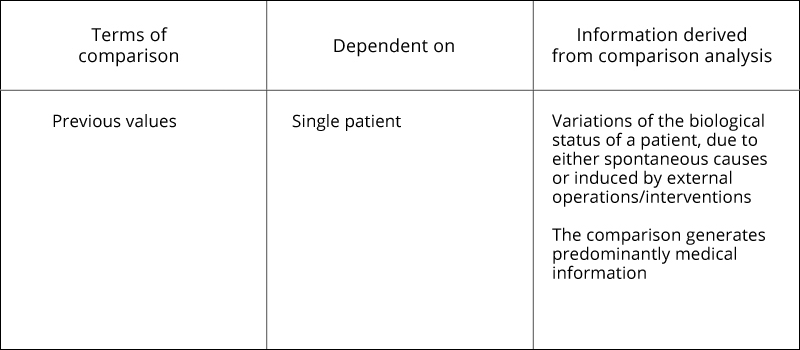
<
>