Methods for Protein Separation
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
Protein Separation Methods


Electrophoresis
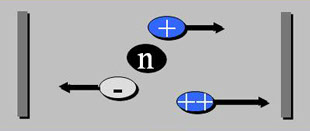
(+) anode
(-) cathode
Analytical separation technique based on the migration of charged molecules under the influence of an electric field.
Electrophoresis

Classification of electrophoretic techniques
Electrophoretic techniques may be classified as follows:
• electrophoresis without carrier molecules: ions migrate within a buffer solution
• electrophoresis with carrier molecules: the most commonly used method, in which ions migrate on supports made of paper, gels or polymers.
• classical electrophoresis (i.e. serum protein separation)
• capillary electrophoresis

ELECTROPHORESIS
Proteins
Their charge depends on:
· Their isoelectric point
the pH value at which a protein is electrically neutral (net electrical charge=0)
· The pH value of the buffer/medium
Electrophoretic separation of proteins can be achieved according to their isoelectric point
KEY PARAMETERS
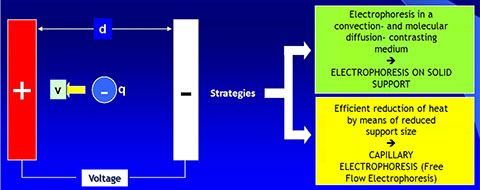
· Electrophoresis is the process by which charged molecules are sorted in an electric field according to their different electrophoretic mobility.
· Factors that affect the electrophoretic mobility of a molecule in an electrical field are: the charge of the given molecule (q), the voltage gradient of the electric field (E) and the frictional resistance of the support matrix (f).
· The force (measured in Newtons) that drives a charged molecule q towards an electrode of opposite charge is equal to the product of q times E (q x E)
KEY PARAMETERS
· The frictional force f tends to slow down the charged particles. This depends on the size and shape of the particles, on the pore size of the matrix support used for electrophoresis as well as the viscosity of the buffer used.
· The speed (v) of a charged molecule that moves within an electrical field is defined by the following equation:


ELECTROPHORESIS – general principles
According to Joule's Law, a current I flowing through an electric circuit with a resistance R generates power (released as heat), given by the following equation:
W = VI = I2R
what is the cause of heat production in such a system?
a) Convection currents
b)Increased molecule diffusion
c) …
}
Negative effect on the separation of molecules - Resolution -
ELECTROPHORESIS
Migration speed
Variables affecting the speed of migration are:
- electric field E= V/d;
- particle charge q;
- friction coefficient f
- (![]() size of molecules)
size of molecules)
v = E q/ f
The speed of migration is thus given by the difference of potential and by the charge/radius ratio (q/r).
Electrophoretic mobility is defined by the following equation: μ=v/E (cm2/volt x sec)

Factors influencing the speed of migration
· SAMPLE
Charge
Size
Shape
· BUFFER
Concentration (ionic strength)
pH
· SUPPORT
Adsorbtion
Molecular filtration
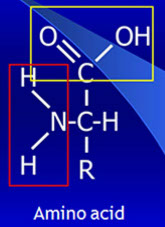
In a basic solution, the carboxyl group behaves as follows:
COOH + OH- → COO- + H2O
In a basic solution, the carboxyl group behaves as follows:
In a slightly acidic solution, the amino group behaves as follows:
NH2 + H+ → NH3+
NH3+ ions will confer a net positive charge to the molecule.
Electrophoresis of serum proteins employs alkaline buffers. At this pH, polypeptides acquire a negative charge which solubilizes them without the need of further denaturation.
ELECTROPHORESIS
Types of separable molecules
· amino acids
· peptides
· PROTEINS
· nucleic acids
· inorganic ions
· organic acids and alkalis
· whole cells
Indications for the Request of Serum Proteins Electrophoresis
Document of the SIBioC PROTEINS Study Group - Approved by the SIMeL PROTEINS Study Group
APPLICATIONS IN CLINICAL BIOCHEMISTRY
Detection of alterations within protein electrophoretic patterns ???
Detection of
monoclonal components

BRIEF HISTORY
1937 Free phase serum protein electrophoresis
Blessum C .Hirumi E .Bullard W. Overview of clinical protein electrophoresis: past and present.
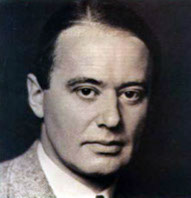
Arne Tiselius, winner of the
Nobel prize for Chemistry in 1948
Serum proteins electrophoresis
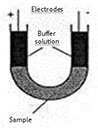
Tiselius first developed the free-phase technique. Protein migration occurred in a buffer solution within a U-shaped tube. A difference of potential was then applied to both ends of the tube. The complex, tedious and poorly standardized technique was replaced by zone electrophoresis on solid support.

SUPPORTS
Non-sifting
(Paper), cellulose acetate
Molecular sieve effect
Polyacrylammide gel (PAG)
Agarose

Electrophoresis in Laboratory Medicine
Different types of electrophoretic techniques
· Zone Electrophoresis · charge/radius
(agarose gel, cellulose acetate, polyacrylamide gel (PAG)) ratio
· SDS-PAGE · Only size
· Isoelectric Focusing · Only charge (pI)
· Two-dimensional electrophoresis · Charge and size
separately
Electrophoresis in Laboratory Medicine
Applications
· Serum and urinary proteins
· Immunofixation
· Haemoglobins
· Carbohydrate-deficient Transferrin (CDT)
· Isoelectric focusing of cerebrospinal fluid IgGs
· Isoenzymes
· Lipoproteins
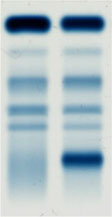
Zone electrophoresis
Following electrophoretic separation, different and distinct "zones" composed of migrating proteins appear on the electrophoretic lane.

}
γ
F. Aguzzi, D. Fenili, N. Montalbetti, C. Petrini, F. Salvatore, M. Tarantino
Official Recommendations of the SIBioC Committee - Serum protein electrophoresis 1°Biochimica Clinica 9; 1985
Zone Electrophoresis
Indications
Detection, monitoring and quantification of monoclonal components
Detection of genetic variants of a protein (i.e.α1-antitrypsin)
Detection of induced protein variants (i.e. bisalbuminemia)
Evaluation of changes within the global protein profile
Dysproteinemias may occur in the presence of normal proteinemia. Total protein concentration may therefore display normal proteinemia or either hypo- or -hyper proteinemia.
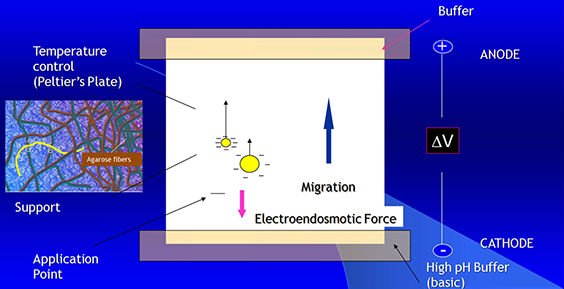
Zone electrophoresis on a solid support
Principles
Signal detection: staining and densitometric quantification (densitometric scanner).
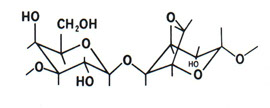
D-galactose
and
3,6 anhydrous L-galactose polymers

State transition
(macroreticular formation by means of hydrogen bonds)
Transition to gel-sol upon heating
Modification of agarose structure upon addition of CH3 groups on OH groups, which occur when temperatures drop to lower transition values (low melting agarose)
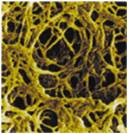
Polymerized agarose reticular network
“molecular sieve”
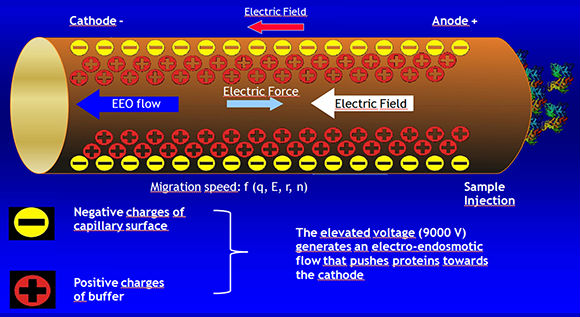
Electroendosmosis
· The porous support medium used in electrophoresis contains groups that acquire a charge relatively to the buffer. This generates a flow of the liquid towards the electrodes.
· In the case of serum proteins, at a pH of 8-9, a strong electroendosmotic effect may be seen with gammaglobulins. These proteins acquire a negative charge (due to the entrapment of H3O ions from water), which implies they do not migrate towards the anode but in the opposite direction (towards the cathode).
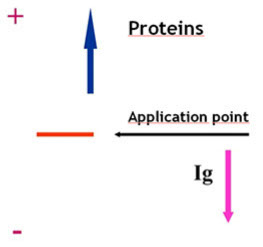


Considerations on electrophoresis
· Increased ionic strength (equivalent to increased sample concentration)determines an increase in current intensity, and consequently increases the Joule heating effect.
· The optimal ionic strength is a good compromise between the length of the run, band resolution and heat production
· Buffers used in the lab range between 0.05 and 0.1
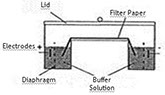
Electrophoresis on solid support mediums
Support medium
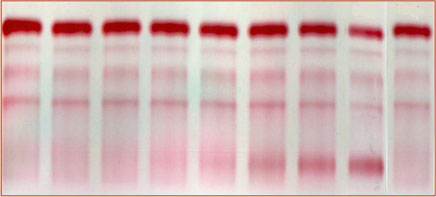
Dry cellulose acetate
Staining method: Ponceau Red or Ponceau S stain
Electrophoresis on solid support mediums
Support mediums
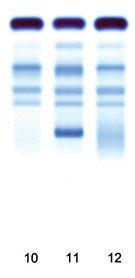
Agarose gel
Staining method: Acid Blue
Cellulose acetate
· Used in routine clinical tests
serum proteins
lipoproteins
detection of abnormal immunoglobulins
· Modest resolution; limited time, low costs
· Nowadays replaced by agarose gel supports
Electrophoresis on solid support mediums
Staining methods
M.C. IgGK: 11 g/L
1:80= 0.138 g\L
1:300 = 0.037 g\L
1:375 = 0.029 g\L


PONCEAU RED OR PONCEAU S STAIN
AMIDO BLACK
ACID VIOLET
Electrophoresis on solid support mediums
Agarose vs Acetate
· Higher resolution
· Does not require transparentization
· Acid Blue staining is more sensitive than Ponceau stain
· Greater versatility of the support in subsequent passages (such as enzymatic reactions)
Agarose supports supply greater sensitivity, versatility and resolution compared to cellulose acetate supports
Electrophoresis on solid support mediums
Limits of the methods
Quantification
· Different affinity of proteins for a specific stain
· Limited linear interval of the densitometric scanning
Automation
· Semi-automatic technique
Zone Electrophoresis
Free zone electrophoresis
Free Zone Electrophoresis
Capillary electrophoresis…
Capillary electrophoresis (mono-capillary instruments) has been used in a variety of applications in research laboratories since the '80s.
A more appropriate technique for routine analyses was developed approximately ten years later, with the implementation of the following aspects:
-The use of more than one capillary
-Faster migration
-Positive identification of samples
-Complete automation
Capillary electrophoresis
Capillary electrophoresis instruments are liquid-based systems composed of two buffer solutions, one capillary with a cooling device, a high-tension energy generator, a system for sample application and a detector.

UV, UV-DAD, fluorimetric, elettrochemical and mass spectometry detectors are available for this technique.
Versions of Capillary Electrophoresis (CE)
Capillary electrophoresis comprises a family of techniques with quite different operative and separative characteristics:
· capillary zone elctrophoresis (CZE): it is the most simple form of CE. The molecules are separated according to differences in mass/charge ratios.
· High Pressure Capillary Electrophoresis (HPCE): an evolved version of traditional CE
· Gel capillary electrophoresis: an adapted version of the traditional gel electrophoresis. The run is operated in a capillary filled with an anti-convection medium such as acrylamide
· Isoelectric Focusing (IEF): molecules migrate in a buffer with a pH gradient. Charged molecules migrate in the electric field until they reach a pH that is equivalent to their IP.
· Isotacophoresis (ITP): particles migrate in the electric field at the same speed
· Micellar electrocinetic capillary chromatography (MECC): the addition of micelle-forming substances enables the separation of neutral molecules (such as phenols)
Free-Zone Electrophoresis
Endosmotic Flow
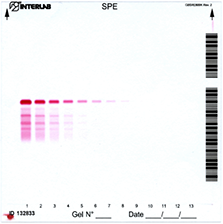
Detection of monoclonal components
Brief History
Immunoelectrophoresis
· Immunoelectrophoresis (IEP) was first introduced by Grabar and Williams. Since then, this technique has enabled the identification of more than 50 serum proteins.
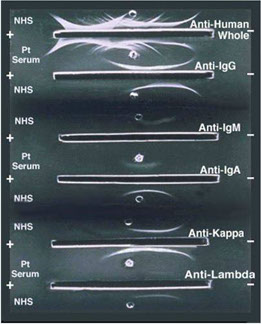

IMMUNOFIXATION (IFE)
SEPARATIVE PHASE
SOLID PHASE ELETTROPHORESIS
+
Anti-
γ α μ δ ε κ λ antibodies
IMMUNOLOGIC PHASE
Sensitivity: 0.005 g/L

IMMUNOFIXATION
Alper CA, Johnson AM. Immunofixation electrophoresis: a technique for the study of protein polymorphism. Vox Sanguinis 1968: 17: 445-52
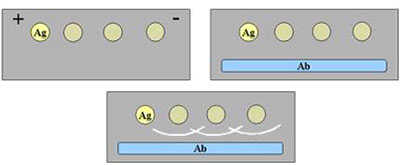
elettrophoresis
Incubation with specific antiserums
Washes
Staining
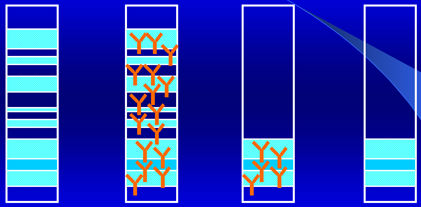
Electrophoresis on SDS-agarose gel


“Sieve” effect in a uniform gel
When a continuous current is applied to a buffer solution with a determined pH, an electric field is generated. Proteins contained in the buffer will move towards one of the electrodes. Speed and direction of each single protein are determined by the surface charge and by the size of the molecule.
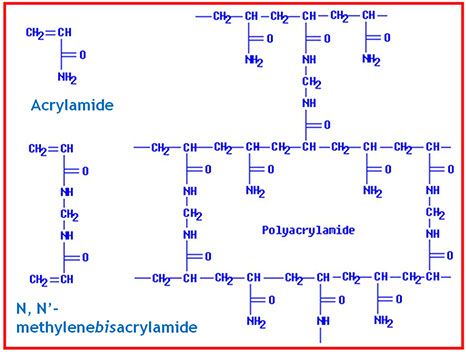
Polyacrilamide (PAGE) is produced from the radical polymerization reaction of acrylamide and a reticulating agent, the "Bis"
PAGE: work-flow
· Gel preparation (“casting”)
- continuous
- discontinuous (“disc”)
- gradient
· Sample preparation
· Electrophoretic run
· Detection
· (Recovery)
POLYACRYLAMIDE GEL ELECTROPHORESIS
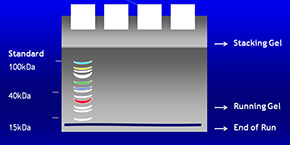
Proteins migrate through the gel according to their molecular weight. Low molecular weight proteins migrate at higher speeds.
The use of a standard molecular weight marker solution containing a mixture of proteins with known molecular weights are used to determine unknown protein molecular weights within the sample of interest.
Fractions obtained after proteins are separated by migration are easily detectable by Ponceau Red staining. The anionic stain reacts with the amide groups of the proteins in acid solutions.
WESTERN BLOT
Analytic technique for separating proteins according to their molecular weights by means of a denaturating polyacrylamide gel. This technique enables the detection of a specific protein (be it the mature form or the precursor) within a given sample.

.jpg)
Protein composed by two subunits, held together by disulfide bonds
Denatured protein. Negatively charged SDS molecules wrap the entire surface of the protein.
Discontinuous PAGE gel system
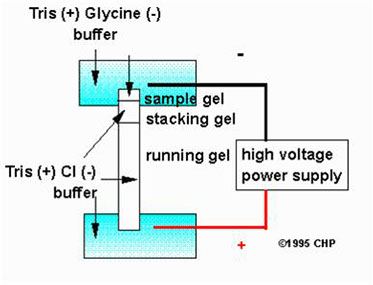
A “stacking” gel is poured on top of the running gel. It is characterized by:
· wider pores
· lower pH
pH 6.8
pH 8.8

Gel in a concentration gradient
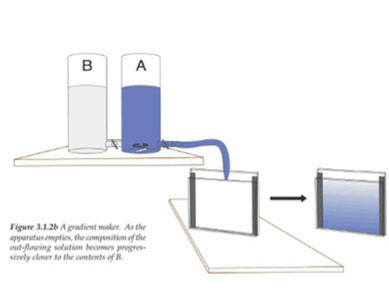
increasing T %
BLOTTING
Process by which macromolecules are transferred and immobilized on a membrane
GEL
MEMBRANE

Blotting: advantages
· The immobilization membrane (nitrocellulose, nylon, PVDF (polyvinylidene difluoride)…) avoids diffusion
· Faster detection compared to gels
BLOTTING
· Southern: DNA
- by Edward Southern, 1975
· Northern: RNA
· Western: proteins (Towbin, 1979)
· Eastern: ? ? ?
Eastern: this is the direct opposite to Western Blotting where you inadvertently connect the power the wrong way round on your gel tank and transfer the proteins from the gel to the tank buffer. Not a recommended protocol."
BLOTTING
· Diffusion
· Capillary
· Vacuum
· Electroblotting (especially for proteins)
Southern Blot (by capillary action)
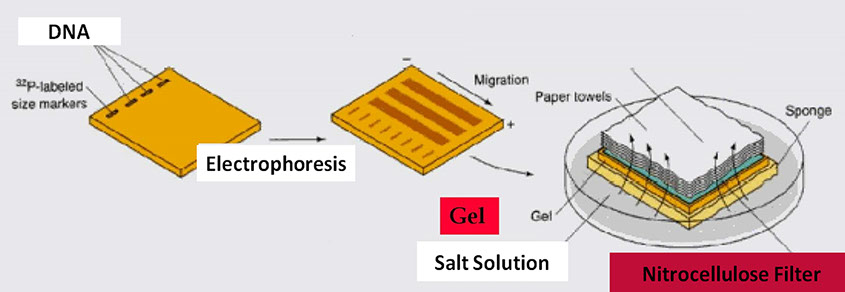
ELECTROBLOTTING
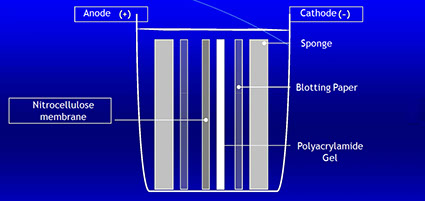
Blotting is a technique for the transfer of proteins from the polyacrylamide gel to a solid support, such as a nitrocellulose membranes, nylon membranes or paper. The gel and the membrane are placed so that negatively charged proteins will migrate from the cathode to the anode and bind to the solid support. The transfer is performed in a wet or semi-dry chamber at constant voltage.
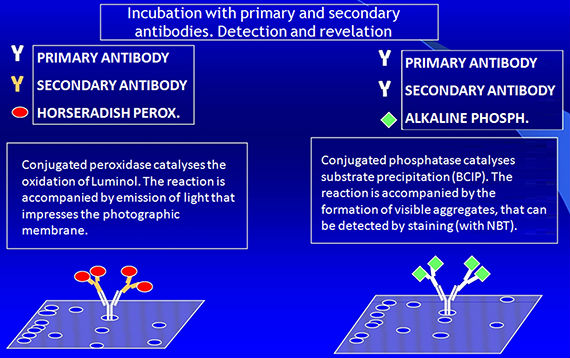
Immunoblotting: detection
· Enzyme-labeled secondary antibodies
· Fluorescence or chemiluminescence (ECL)
· Biotynilated secondary antibodies
· Gold-labeled secondary antibodies
· Radioactive Protein A
· Lectins (for glycoproteins)
PROTEIN DETECTION AND IDENTIFICATION
Isoelectric focusing (IEF)
IEF is an electrophoretic method for separating proteins based on their Isoelectric Points (pI)
In an electric field with a pH gradient, proteins will migrate until they reach a pH value where their net charge is zero: this depends on their pIs

ISOELECTRIC FOCUSING
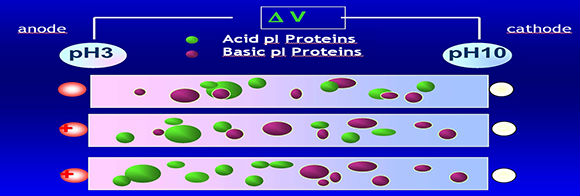
pH gradient in isoelectric focusing
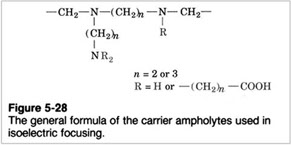
The pH gradient is obtained by adding "ampholytes" to the gel. Ampholytes are complex mixtures of synthetically derived polyaminocarboxilic acids.
These low molecular weight molecules behave as acids or bases. In the electric field they will move between the electrodes until they reach the position corresponding to their pI, giving rise to a gradient.

A.No voltage
B.Ampholytes and proteins move according to their charge when a current is applied
C.Proteins are “focused” at the point in the pH gradient corresponding to their pIs.
Two-Dimensional Gel Electrophoresis (2-DE)
· “First dimension”: IEF
Separates proteins according to their pIs (isoelectric points)
· “Second dimension”: SDS-PAGE
Separates proteins according to their molecular weight
PROTEIN MAP

ID=IEF. Proteins are adsorbed on dry gel strips with immobilized pH gradients. Upon rehydration, proteins will migrate in the electric field: acid proteins on the basic side will dissociate and acquire a negative charge. Due to the presence of the electric field, these proteins will migrate towards the positive electrode (acidic side of the gel).
2D=SDS-PAGE
Proteomics: two-dimensional electrophoresis coupled with mass spectrometry
Two-dimensional gel electrophoresis (2D-gel) with mass spectrometry can be used for proteome analysis in order to identify specific proteins of interest.
Currently available technical apparatuses enable to reproducibly separate proteins with only 1 pH unit difference in a 24 cm run.

Mass spectrometry (MS-Maldi-TOF) is the most commonly used method for analyzing protein aminoacid composition.
Mass Spectrometry
· Ions are separated in a gaseous phase according to their charge/mass ratio
· Steps of the analysis
sample application
ionization (MALDI or ESI)
separation (TOF or ION-TRAP)
detection
elaboration
Mass Spectrometry
MS-MALDI TOF
- Sample molecules are converted to gaseous ions
- Ions are accelerated in either a magnetic or an electric field. The TOF of ions with different masses is measured at a given distance
- Ions are separated by a charge-mass analyzer (m/z)
- The mass/charge ratio of each ionized species is determined by the detector
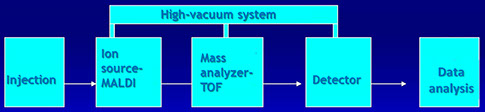
TOF=Time of Flight
Time needed by ions of different masses to travel the length of the flight tube
MALDI-TOF
(Matrix-Assisted Laser Desorption Ionization)
Time of Flight mass spectrometry
Electrophoretic Analysis
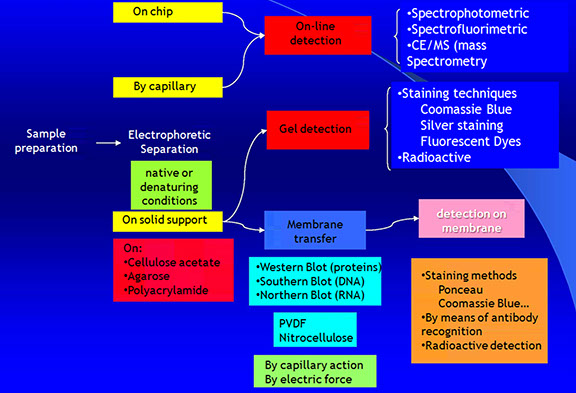
<
>