Bence-Jones Proteinuria
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
69
70
71
72
73
74
75
76
Bence-Jones Proteinuria:
evaluation of the best analytical techniques
Principles of Pathophysiology
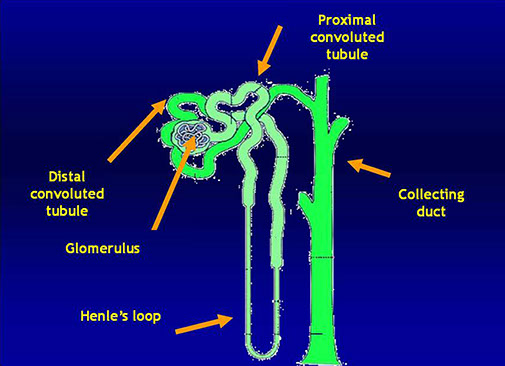
Kidney anatomy

The nephron: the functional unit of the kidney
The blood circuit of the Nephron
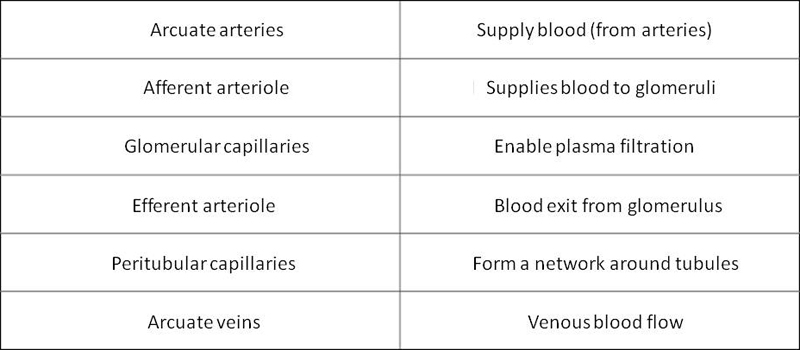
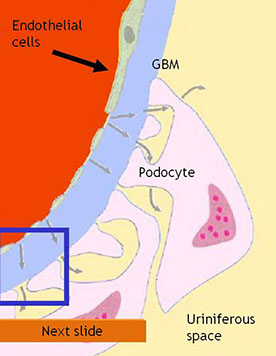
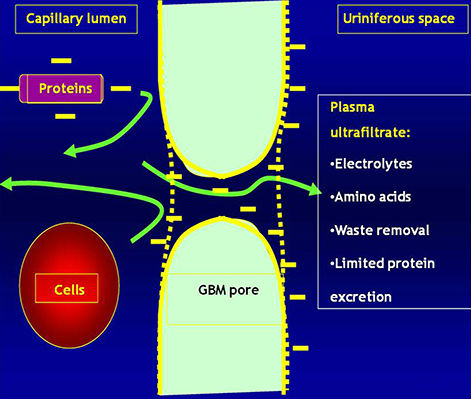
The glomerulus
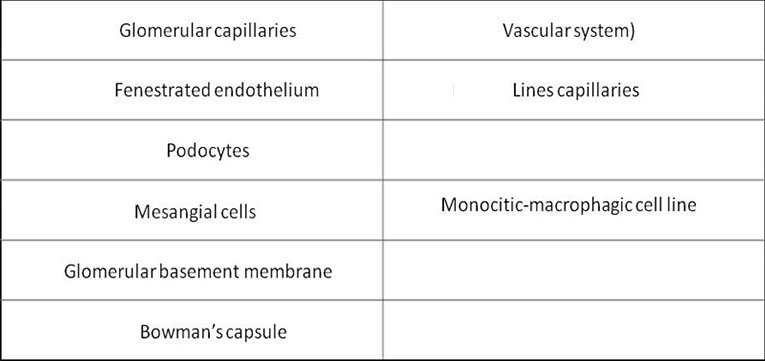

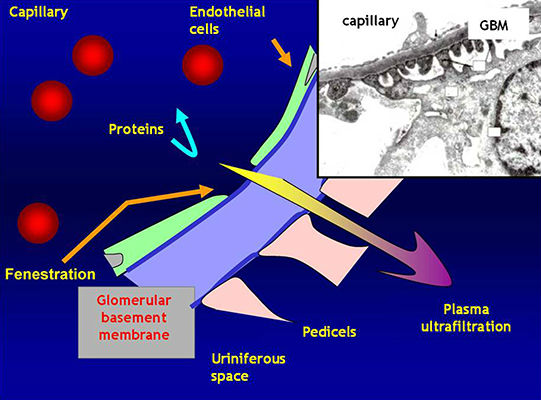
The glomerulus
· Plasma is filtered through the Glomerular Basement Membrane (GBM)
· The glomerular endothelium is fenestrated
· The pedicels of podocyte are in contact with the GBM
· Filtered fluid from plasma filtration (ultrafiltrate) flows into Bowman's space
Tubules


Nephron and protein physiology
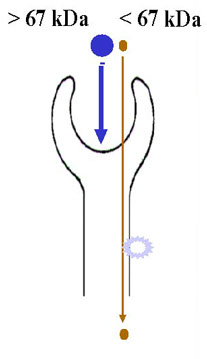
albumin
transferrin
immunoglobulins
microproteins
albumin
transferrin
immunoglobulins
microproteins
microproteins
Glomerular filtration
Tubular re-absorption
Tubular secretion
(Tamm-Horsfall protein)
Urine: final product composed of 1/3 albumin and
2/3 globulins
The final urine product is the combined result of glomerular ultrafiltration and tubular reabsorption
Upon physiological conditions, these processes prevent undesirable protein losses in the urine

Diagnostic relevance of urinary protein electrophoresis
· Detection and characterization of monoclonal free light chains (or Bence-Jones Proteins-BJP) and monoclonal components in the urine (assessment of disease progression in monoclonal gammopathies, prevention of problems relatively to nephrotoxic actions of BJP).
· Detection and characterization of high and/or low molecular weight proteins for the assessment of renal damage (pre-renal, glomerular, tubular, post-renal).

Increased proteinuria: causes
· Physiological: intermittent (orthostatic, postprandial, effort, stress)
· Pre-renal: hypertension, hyperthermia, cardiac or liver affections, ...? Elevated protein concentration (proteinuria overload)
· Renal: glomerular or tubular (or both) injuries
· Post-renal: urogenital tract damage and injuries





Mixed Proteinuria


Bence-Jones Protein:
the first tumor marker in Medical history
· Henry Bence Jones (1813-1873) was a physician at St. George's Hospital in London.
· In 1845, he received a urine sample from a patient suffering from "mollities ossium". He then isolated a protein that, in contrast to albumin, was soluble in boiling nitric acid. In 1847, he reported the discovery of this new protein [Phil Trans R Soc Lond 1848; 138: 55].
· The metabolic origin of this protein, the nature of the neoplastic disease (multiple myeloma) along with the association between this affection and the detection of this protein in the urine, were clarified later.
Bence-Jones Proteins
Crystallographic structure of a Light Chain
Monoclonal Free Light Chains
Bence-Jones Proteins
· Plasma cells normally produce excess light chains that do not bind heavy chains.
· Modest amounts of these polyclonal light chains are filtered (3 to 4 mg/day) through the kidney and partially reabsorbed by the proximal convoluted tubule, where low molecular weight proteins are catabolized.
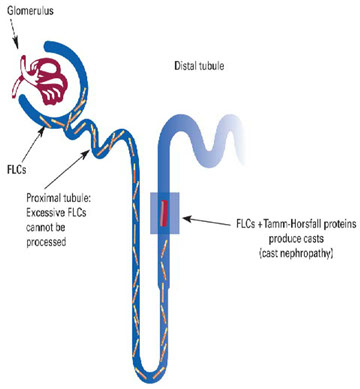

Bence-Jones Proteins


Bence-Jones Proteins
· Excess production of this protein (as in neoplastic diseases) can cause renal tubule damage. In time, this condition will lead to various degrees of renal impairment.
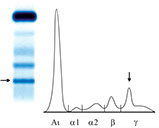
· BJP is detectable by means of urinary electrophoretic analysis. It appears as a band of variable migration due to its great variability. Indeed, BJP exists in multimeric forms and can be found as monomers with molecular weight of 22 kDa, dimers of approximately 44 kDa and fragments weighing from 5-18 kDa.
Bence-Jones Proteins
Urinary E.F.
Presence of Bence-Jones Protein.
The different localization of the proteins is indicative of various degrees of polymerization.



PATHOLOGIES ASSOCIATED TO BJ PROTEIN
BJP can be detected in different pathological situations:
multiple myeloma
Waldenström macroglobulinemia
amyloidosis
light chain deposition disease.
The presence of BJP is less frequently associated with lymphomas and chronic lymphocytic leukemia. It rarely occurs in association with lymphoproliferative malignancies.


APPROPRIATENESS
A medical examination is deemed appropriate when the test has the ability to change outcomes or medical decisions: Learning Organization
· Appropriateness is a relatively young term in health care environments, and even more from the legal point of view (with regards to Italy). Nevertheless, it is quickly acquiring greater importance as a guiding principle and a priority within Health Services, especially in relation to to streamlining and optimization of health costs expenses.
· The term "appropriateness" was first introduced in the Italian healthcare system in 1998. Quoting the National Health Plan 1998-2000: "The wide variability in (medical care) responses is due to appropriateness issues concerning the unequal use of resources as well as the potential access to the use of health services. "
APPROPRIATENESS
APPROPRIATENESS IMPLIES
· doing what is right
· at the right time
· in the best way
· for those who need it
· with clinical expertise (clinician, clinical pathologist, patient)

Bence-Jones Proteins: indications
Patients affected by serum monoclonal gammopathy:
upon detection and at every subsequent check-up
Clinical or laboratory suspicion
(eg. unexpected hypogammaglobulinemia in adult subjects) of light chain disease
Clinical suspicion of amyloidosis or light chain deposition disease


Analytical aspects


Sample
URINE: concentrated or unconcentrated samples
Sample
- BJP is easily degraded by bacteria in the bladder. For qualitative analysis, it is important to use fresh urine (with the minimum bladder incubation).
- Fresh urine samples are preferred (second-morning-void urine samples, collected between 6 and 9 pm). For 24 hour sample collection, sodium azide should be added (1%) in order to stop bacterial growth.
· Absolute urine can be used if the detection method is sufficiently sensitive. If, however, BJP detection with highly sensitive methods is clinically required, urine samples must be concentrated.
Sample concentration
Concentrate=(Chemistry) to make or become denser or purer by the removal of certain elements, especially the solvent of a solution.
Collins English Dictionary – Complete and Unabridged © HarperCollins Publishers 1991, 1994, 1998, 2000, 2003
Methods for concentrating biological fluids
DIALYSIS
PRECIPITATION CHEMISTRY
EVAPORATION
DRYING
LYOPHILIZATION
ULTRAFILTRATION
Limits
The aforementioned techniques may exhibit a variety of difficulties, summarized as follows:
ELEVATED COSTS
LONG OPERATING TIMES
CHEMICAL OR PHYSICOCHEMICAL VARIATIONS OF COMPONENTS
PERTURBATION OF BIOLOGICAL FLUIDS
LOSS OF "BIOLOGICAL ACTIVITIES"
LOSS OF MOLECULES DUE TO MEMBRANE PORE DIMENSIONS
Technical issues: how to concentrate samples?
· Determine the sensitivity of the system used
· Concentration should not exceed 10X.
· SIBioC guidelines concerning the sensitivity of methods for BJP detection: at least 10 mg/L
· Sensitivity of agarose methods: approximately 6-12 mg/L
· Select porosity equal to or less than 10,000 D
· Concentrate at a fixed temperature
Technical issues in static ultrafiltration ? Limits in solute recovery



· Qualitative measures
· Loss of solute due to non-specific binding
· Loss of solute due to pore size
· Elevated concentrations of little use for low recovery
Ultrafiltration concentrators by centrifugation (Vivaspin)
The advantages of concentration by ultracentrifugation are:
· Molecular weight cut-off of 5 kDa
· Absence of concentration polarization
· Reduction of dipole-dipole retention
· Significant reduction of diffusion phenomena and greater solute recovery index.

Principles of adsorption techniques
The binding of gaseous or liquid molecules (or particles) to a solid surface.
The term "surface" includes external as well as inner canaliculi, capillaries, fractures, etc. of a given material (quoted from the Treccani Encyclopedia)

Adsorbents
· Granular silica gel: adsorbent surface.



Concentration
Metal alloy base container for the distribution of biological samples (urine, cerebrospinal fluid, etc.) in defined volumes (120 uL)


Constant T silica gel CONCENTRATOR

Detection and characterization of Bence-Jones Proteins
Screening implies an action on a particular population in order to identify an unknown disease by means of simple and rapid testing.
DETECTION
Method
The most frequently used approach in clinical laboratories is to perform an initial electrophoretic screening, followed by subsequent immunofixation, order to verify the presence of free light chains.

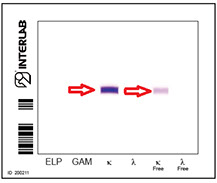
Method detection limits Electrophoresis<sensitive IEF
Sensitivity 0.029 g\L
Sensitivity 0.005 g/L


ACID VIOLET
ACID VIOLET
Bence Jones Protein

Immunofixation is considered the "gold standard" method. In the medical field, this technical term refers to either a substance, a drug, a test type, a medical or surgical treatment considered as the most reliable approach.
In our case, this test amplifies the sensitivity and enables to detection small but
clinically significant amounts of BJP.
IMMUNOFIXATION
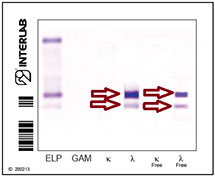
electrophoresis
incubation with
monospecific antisera
PENTAVALENT IFE

· Concentrate urine samples using concentrators with 5 kD molecular weight cut-offs
· Agarose gel IFE PENTA 6/12 and 2 strips of immunofixation buffer are used for each run.
· Dispense 12 μl of concentrated urine in double (two wells for each sample)
· 5 min deposition step at 20°C
· Electrophoretic migration in alkaline pH (prog.BJ)
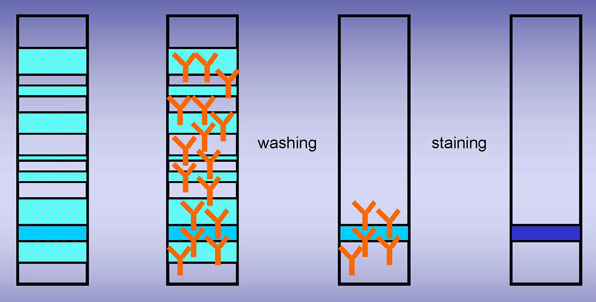
· Immunoprecipitate with ELP and antiserum Pentad (Ig γ α μ and κ and λ chains).
· Remove precipitated proteins
· Stain (crystal violet)
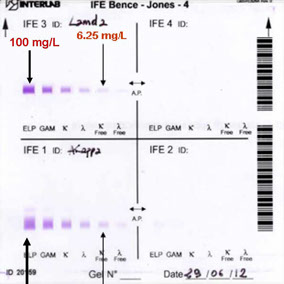
The first lane of each electrophoretic run shows total proteins. A pentavalent antiserum (anti- Ig γ α μ and κ and λ chains) was applied to the second lane.
EF Pattern 1
Presence of proteinuria
No M.C.
EF Pattern 2
Absence of proteinuria
M.C. positive
EF Pattern 3
Alb. + traces
No M.C. detected by EP
EF Pattern 4
Traces of Alb.
No M.C. detected by EP




Histograms showing frequency distributions of relationships between B.J.-IFE and URINE-HRE
Histograms showing frequency distributions of relationships between B.J.-IFE and PENTA-IFE

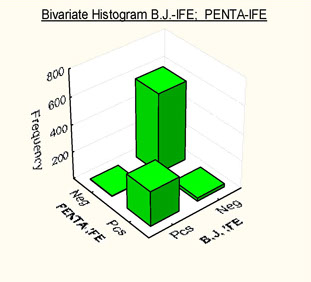
DATA BY Basile U. et al
DATA BY Basile U. et al
PENTA IFE
· MC detection in urine has highlighted how even the best protein electrophoresis methods may occasionally be unable to detect the presence of monoclonal components.
· PENTA IFE has shown "excellent sensitivity", practical performance of the test as well as cost containment, thus suggesting the use of this technique as screening tool for implementing monoclonal component detection in urine.
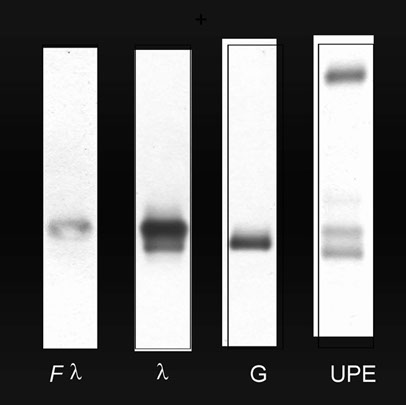
New BIOCI Method – BJTS Bence-Jones Tubular Screening for the detection of BJP
· A mixture of anti-Kappa and Lambda chains antiserum (free and bound) together with anti-alpha1-microglobulin IFE on agarose gel performed as single electrophoretic run/sample.
· Possibility of screening unconcentrated urine by crystal violet staining. Cost- and time- effective.
Data by Vernocchi A.
Sensitivity tests
Interlab agarose gel tested with BIOCI anti- Kappa and Lambda Light Chains antibodies
Method
· Staining the immunofixed gel with colloidal stains (such as gold) enables to reach adequate levels of sensitivity (< 10 mg/L) without the need of sample concentration.
· The use of techniques providing a combination of high sensitivity and good resolution implies the possibility of frequent detection of a series of multiple bands (mainly with the κ antiserum, but also with the λ antiserum). These bands have no clinical significance, but may be confused with BJP.
· They can be distinguished from BJP due to their typical and repetitive pattern, composed of regularly spaced bands.
Method


Bence-Jones Protein:
a suggested protocol for detection and typing of BJP
Second morning void.
CAP 24h


Recommended sensitivity: 10mg/L.

In the presence of monoclonal gammopathy and/or BJP, the most important parameter is the progression of the clinical picture.
The quantitative determination of both, the MC and/or the BJP, is therefore required.
Bence-Jones Proteins:
Proposed approach for BJP quantification
24h urine protein quantification for proteinuria
(specific method such as Pyrogallol Red).

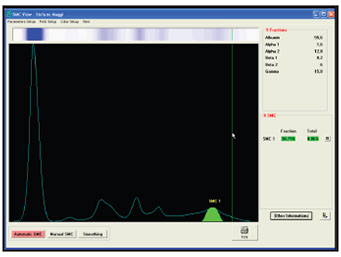
EP (zone or high resolution EP)
Quantification of band intensity (in terms of percentage)
Expression of the % value in g/L .

Example: Quantification

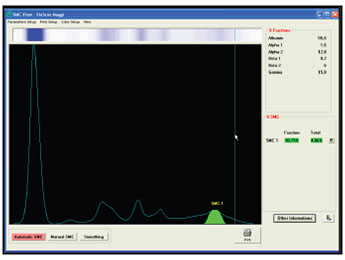
MC (g/L) = % value * Proteinuria (g/L)
Limits of detection
Available techniques for urine total protein quantification exhibit difficulties in microproteins and BJP detection. TP values of samples with BJP may thus be inaccurate.
Stain affinity may vary according to different proteins. A given amount of protein may not necessarily correspond to a given intensity and it has not been proven that equal intensities correspond to equal amounts of protein.
A split BJP band can occasionally be observed. This may by difficult to isolate from the rest of the bands on the lane as well as to evaluate densitometrically. Further evolutions of the capillary-based techniques may solve the problem.
Despite its limitations, this technique is the only method currently available for the detection and the quantification of BJP (as suggested by the CAP and the SIBioC guidelines).
Bence-Jones Proteins:
Immunometric quantification of total k and l chains
Better repeatability and standardization.
It also enables to measure out small entity BJPs that co-migrate with other proteins that might also be present.
Nevertheless…
The calibration curve is achieved by means of polyclonal kappa and lambda chains. This does not guarantee a precise quantification of monoclonal chains. Parallelism between calibrator and the antigen within the sample, an essential requirement for an immunological test, is not satisfied.
Poor repeatability and imprecision in the presence of aggregates.
QUANTIFICATION
Conclusions
· BJP quantification is crucial for the differential diagnosis of conditions associated with the presence of monoclonal components (MC) as well as for patient monitoring .
· However, the aforementioned problems can not be solved with currently available laboratory techniques.
Limitations of the procedure
· Despite the aforementioned issues, the CAP densitometric procedure is the only available technique for BJP quantification. No alternatives are currently available.
· Performing follow-ups in the same laboratory is, however, recommended in order to minimize analytical variability.
· It is worth mentioning that current studies concerning the application of capillary electrophoresis could lead to a solution of the problem.

• Bence Jones Protein can be found in very elevated amounts. This may determine antigen-eccess effects.
• The BJP often forms aggregates of variable sizes . This aspect may determine poor repeatability of the immunometric quantification
The following methods should be discouraged:
• Methods for total protein quantification (precipitating or dye-binding techniques) due to their poor sensitivity and accuracy
• Dip-sticks for standard urine testing. Such methods are based on protein error and are only sensitive to albumin detection
• Heat test, of mere historical significance.
Guidelines for the diagnosis and monitoring of MM:
an update
· Since the publication of the guidelines by the IMWG (2009) concerning the use of FLC assays in this field, many guidelines for the diagnosis and monitoring of MM and related dyscrasias have been updated and developed.
· None of these guidelines (at national or international level) recommended to exclude urinary electrophoresis or urinary immunofixation from the initial diagnostic algorithm.

We realize that the strong pressure to reduce laboratory costs makes our approach difficult to sustain, but we believe that new, less expensive technologies/procedures should be adopted only if they provide optimal clinical efficacy.
“We cannot afford missing the diagnosis of a single patient with malignant monoclonal gammopathy”.

G Merlini et al.. Clin Chem Lab Med 2001;39:1025-28
<
>



