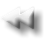National and International Guidelines
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
NATIONAL AND INTERNATIONAL GUIDELINES

Serum FLC ratio is an indicator of monoclonality and a valuable tool to discriminate between monoclonal and polyclonal diseases.
Freelite serum immunoassay
Heavy
chain
Light
chain
Hidden
surface
Lambda
Kappa
Exposed
surface
Free Light Chain Physiology
· 500-1000 mg of Free Light Chains produced per day
- 2/3 Kappa
- 1/3 Lambda
Half-life
- Kappa: 2-4 hours
- Lambda: 3-6 hours
Clearance
- Proximal tubules
- Reticuloendothelial system
The Freelite assay and the International Guidelines
· “serum FLC assay, in combination with serum protein electrophoresis and
immunofixation, yields high sensitivity [...]for the purpose of screening for monoclonal proteins for performing all diagnoses with the exception of AL, the FLC assay can replace 24h urine IFE; for AL screening, however, urine IFE should still be performed, in addition to serum tests, including serum FLC quantification”.
· “the FLC assay is of major prognostic value in virtually every plasma cell disorder, including monoclonal gammopathy of undetermined significance, smoldering myeloma, active myeloma, immunoglobulin light chain amyloidosis and solitary plasmacytoma.”

“Serial FLC ascertainment should be routinely performed in patients with AL amyloidosis and multiple myeloma patients with oligosecretory disease”.
· “It should also be done in all patients who have achieved a CR to determine whether they have attained a stringent CR.”.
· “The serum FLC assay should be measured at diagnosis for all patients with MGUS, smoldering or active multiple myeloma, solitary plasmacytoma and AL amyloidosis ”.
International Myeloma Working Group guidelines for serum-free light chain (FLC) analysis in multiple myeloma and related disorders
There are three major indications:
1 - DIAGNOSIS of AL-Amyloidosis (in combination with SPE, serum IFE and BJP)
2 - as baseline PROGNOSTIC VALUE in virtually every plasmacell disorder (PCD)
3 - Quantitative MONITORING of patients with AL, oligosecretory myeloma, 2/3 of previously diagnosed NSMM.
Dispenzieri A et al. on behalf of the IMWG, Leukemia 2009,23:215-24
In caso di sospetto di amiloidosi AL



· United States
· January 2011

· These guidelines concern MM, solitary plasmacytoma, AL amyloidosis
· The Free Light Chain assay was first included among the NCCN guidelines in 2010
· It was included within the initial diagnostic panel not as a screening test, but rather suggested as "useful in some circumstances"
· In January 2011, the FLC assay was moved upstream in the algorithm, among the initial tests of the panel
· The FLC assay was then added to the standard test panel for initial diagnosis and did not replace urine tests!

· The 2009 IMWG Guidelines mainly focused on the correct use of the FLC assay itself. No recommendations concerning the use of other tests or suggested algorithms were included in this document.
· The new guidelines provide recommendations on the use of all tests.
· The new recommendations concern multiple myeloma (do not include AL amyloidosis; MGUS, smoldering myeloma and plasmacytoma are only roughed out).
Hevylite Assay
· Katzman et al (Leukemia 2013, 27: 208-212): sHLCr may have prognostic significance for monitoring the evolution of MGUS to MM;
· Astolfi et al (2013) :
· SHLCr sFLCr more sensitive for predicting MM recurrence;
· SHLCr more effective parameter for monitoring follow-up of MM patients;
· SFLCr + sHLCr useful for monitoring patients with MM.
Tests recommended by the Italian National Guidelines
· Test both SERUM and URINE.
· In the presence of clinical suspicion of MC, perform IFE by means of anti-IgA, IgG and IgM antiserums.
· IgD and IgE should be tested if only κ or λ chains are initially detected.
· uBJP performed on urine concentrated 50x-100x.
· Use concentrators with a 5 Kda cut-off.
The sFLC assay is useful for the diagnosis and management of patients affected by low-Secretory Myeloma and AL, or whenever urine specimen are unavailable.
<
>